NURS 6630 Week 2 Discussion Foundational Neuroscience
Agonist-to-antagonist Spectrum of Psychopharmacologic Agents
A medicine or substance that binds to and activates a specific receptor to create a desired biological response is known as an agonist. Exogenous agonists are substances that imitate the activities of endogenous agonists. Endogenous agonists are those created within the body, whereas exogenous agonists are those synthesized outside of the body. Agonists are divided into three types based on their intrinsic efficacy: full agonists, partial agonists, and inverse agonists.
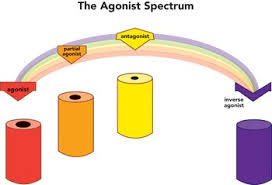
A complete agonist has a high intrinsic effectiveness, which means it activates all receptors to their maximum potential, resulting in the maximum expected response. A partial agonist has a lower intrinsic efficacy than a full agonist, which results in sub-maximal receptor activation and a reduced physiological response. Despite binding to the same receptor location as the agonist, an inverse agonist has a negative intrinsic effectiveness because it causes a physiological response that is antagonistic or opposite to the agonist (Berg & Clarke, 2018). An antagonist, unlike an agonist, has no intrinsic efficacy but has a high affinity for receptors and inhibits the effects of agonists. Depending on the drug’s intrinsic activity and affinity, the agonist-to-antagonist spectrum has consequences for the choice of psychopharmacological drugs.
Comparison Of G Couple Proteins And Ion Gated Channels
G protein-coupled receptors GPCRs and ion gated channels (IGCs) are receptors that are usually activated by stimuli to mediate cellular responses. GPRCs are more numerous than IGCs and function through second messenger networks. The activation of these receptors by ligands enables them to bind to G-proteins, which promote the exchange of GTP for GDP, resulting in a cascade of activities that induce a cellular response (Weis et al., 2018). IGCs are transmembrane proteins with a central pore that opens and closes to regulate ion flow across cell membranes (Phillips et al., 2020). Due to the coupling, GPCRs respond to signals more slowly and are activated by slower neurotransmitters like serotonin. Faster neurotransmitters, such as acetylcholine, activate IGCs, resulting in a faster response.
Role Of Epigenetics In Pharmacologic Action
Epigenetics is increasingly being adopted in the development of therapeutic pharmacological agents. In epigenetics, DNA methylation or histone modification regulate DNA transcription without inflicting any changes to the DNA sequence (Kringel et al., 2021). Through conformational changes in transcription factors, this results in enhanced or decreased transcription of the target genes or exposure of desired regions of the DNA. As a result, drugs targeting specific genes that contribute to the genesis of numerous diseases, including psychiatric illnesses, can be produced. However, the efficacy of these therapeutic epigenetic pharmacologic agents, as well as expected responses, may be dependent on the existence or modification of the target genes among individuals; so, diversity is to be expected.
Impact of the Above Information on Prescription of Medications to Patients
Given the foregoing, it is critical to understand an individual’s genetic background prior to treatment. This is due to the fact that some genes are heritable, which is most typically seen in the medical environment as a positive family history of a particular sickness. As a result, obtaining a complete medical and family history before to treatment is critical. In the case of multiple therapy failures, genetic testing should be considered. Epigenetics is influenced by genetic and environmental variables; thus, medical professionals must be aware of this. Similar conditions in twins, for example, may not respond to treatment in the same way, necessitating the need to tailor care.
NURS 6630 Week 2 Discussion Foundational Neuroscience References
Berg, K., & Clarke, W. (2018). Making Sense of Pharmacology: Inverse Agonism and Functional Selectivity. International Journal Of Neuropsychopharmacology, 21(10), 962-977. https://doi.org/10.1093/ijnp/pyy071
Kringel, D., Malkusch, S., & Lötsch, J. (2021). Drugs and Epigenetic Molecular Functions. A Pharmacological Data Scientometric Analysis.&nb